
Stima del rischio con il metodo SCRAM (SICK) Rev. 1.0 Settembre 2022
ID 15490 | 16.09.2022 / Documento completo (EN) in allegato
Risk assessment and risk reduction for machinery - Part 3: Conducting risk estimation. Scalable risk analysis and evaluation method (SCRAM)
Update Rev. 1.0 2022
- SICK AG - WHITE PAPER 2022-05
….
Introduction
Scope
Machine risk assessment consists of a series of steps used to examine the hazards associated with machines and it consists of two stages, namely risk analysis and risk evaluation, as laid out in ISO 12100:2012. Risk analysis comprises three stages: determining the limits of the machine, identifying hazards, and estimating the risk.
After having completed the hazard identification phase, risk estimation is carried out for each identified hazard and hazardous situation. Risk is defined as a combination of the severity of harm and the probability of occurrence of that harm.
According to ISO 12100:2012, the probability of occurrence of harm can be estimated taking into account the frequency and duration of exposure to the hazard, the probability of occurrence of a hazardous event, and the technical and human possibilities to avoid or limit the harm. The combination of the severity of the possible harm with these three probability parameters will be used to estimate risk values which can then be used for comparison purposes. At the last stage of the assessment process, risk evaluation allows decisions on risk reduction measures to be applied to the machine.
The scope of this white paper is to provide a risk estimation methodology that has proved to be robust, and reliable while preventing errors when estimating risks.
Preface
This white paper is part of a series of papers describing the SICK process of risk assessment in combination with risk reduction:
- Part 1: Defining the scope of the risk assessment
- Part 2: Identifying task/hazard pairs
- Part 3: Conducting risk estimation
- Part 4: Integrating protective devices into (existing) control systems
- Part 5: Implementing emergency operations
- Part 6: Carrying out substantial modifications
Risk reduction process
General
All products and systems include hazards and, therefore, some level of risk. However, the risk associated with those hazards shall be reduced to an acceptable or tolerable levela. The iterative process of risk assessment and risk reduction for each task and hazard combination is essential in achieving acceptable risk (level of risk that is accepted in a given context based on the current values of society).
The objective of risk reduction can be achieved by the elimination of significant hazards, or by reducing each of the two elements (separately or simultaneously) that determine the associated risk:
- Severity of harm from the hazard under consideration
- Probability of occurrence of that harm
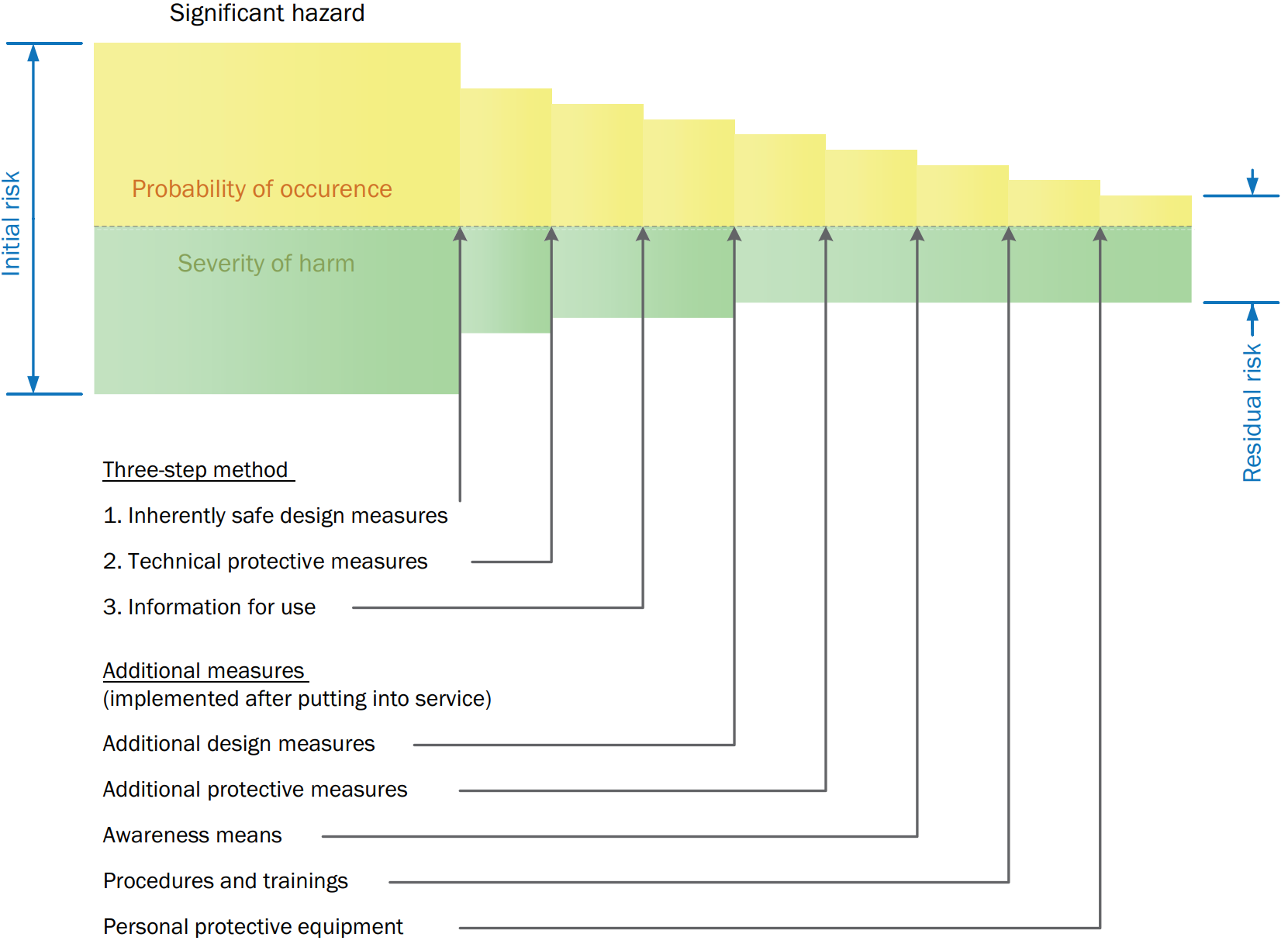
Fig. 1 - Overall risk reduction
a For the purpose of this document, the terms “acceptable risk” and “tolerable risk” are considered to be synonymous.
Three-step method
All risk reduction measures intended for reaching this objective shall be applied in the following sequence, referred to as the threestep method:
STEP 1: INHERENTLY SAFE DESIGN MEASURES
Inherently safe design measures are achieved by avoiding hazards or reducing risks by implementing a suitable choice of design features for the machine itself and/or interaction between the exposed persons and the machine.
Inherently safe design measures are the first and most important step in the risk reduction process. This is because risk reduction measures inherent to the characteristics of the machine are likely to remain effective, whereas experience has shown that even well-designed safeguarding can fail or be defeated and information for use may not be followed.
STEP 2: TECHNICAL PROTECTIVE MEASURES
Guards and protective devices (also known as “safeguarding” or “engineering controls”) shall be used to protect persons whenever an inherently safe design measure does not reasonably make it possible either to remove hazards or to sufficiently reduce risks (for details see chapter “Technical protective measures”). Complementary protective measures involving additional equipment (for example, emergency stop equipment) may also be necessary.
STEP 3: INFORMATION FOR USE
Information for use consists of communication means, such as texts, words, signs, signals, symbols, or diagrams, used separately or in combination to provide information to the user (employer and/or affected persons).
The information shall contain all directions required for safe and intended use of a machine. To achieve this purpose, it shall also inform and warn the user about residual risk.
The information shall indicate, as appropriate, the requirements for additional measures that the user shall implement:
- The possible need for additional guards or protective devices
- The consideration of regular inspections
- The consideration of safe work procedures and training
- The consideration of personal protective equipment
Information for use shall not be a substitute for the correct application of inherently safe design measures, technical protective measures, or complementary protective measures.
[...]
Progressive iterations in risk assessment
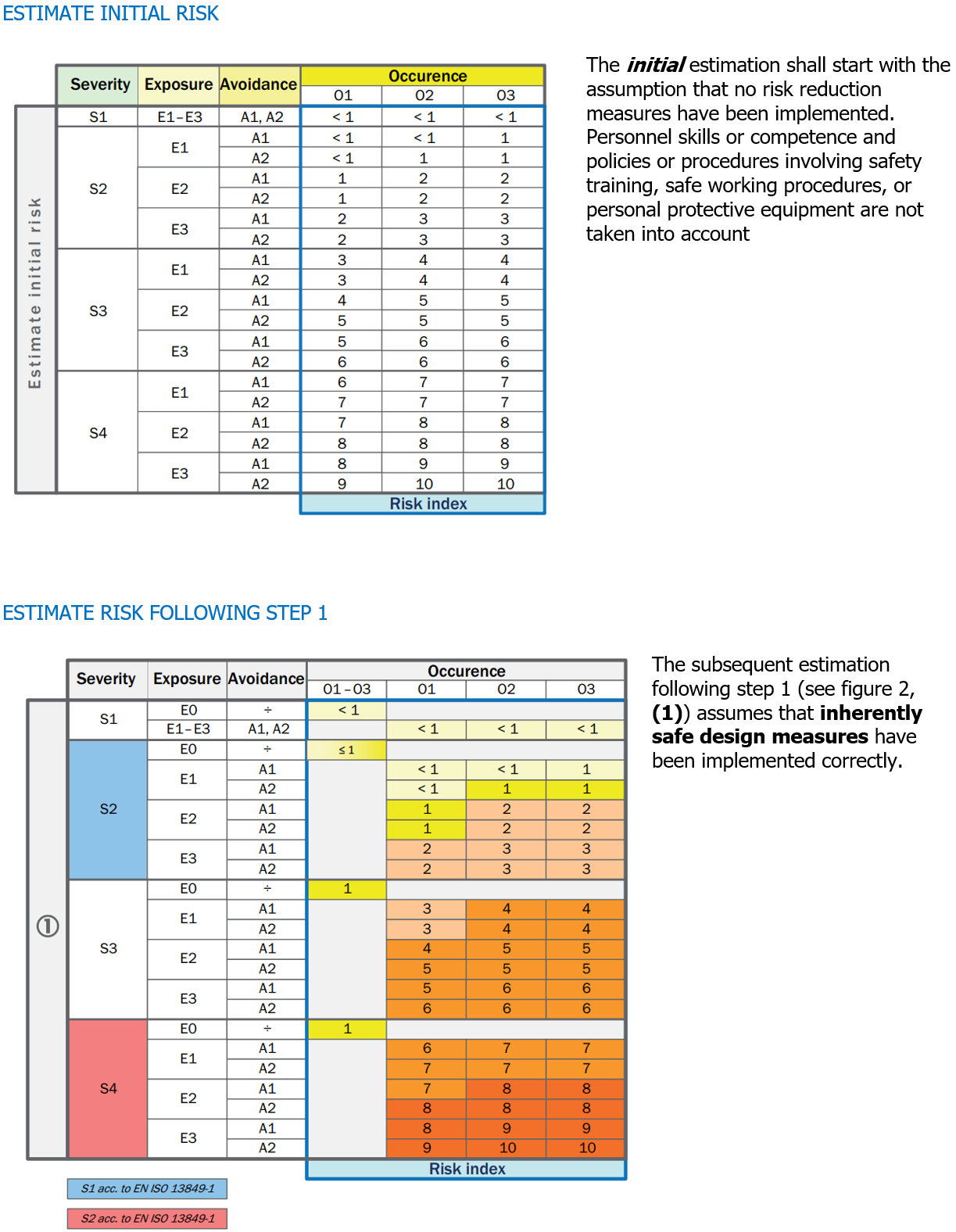
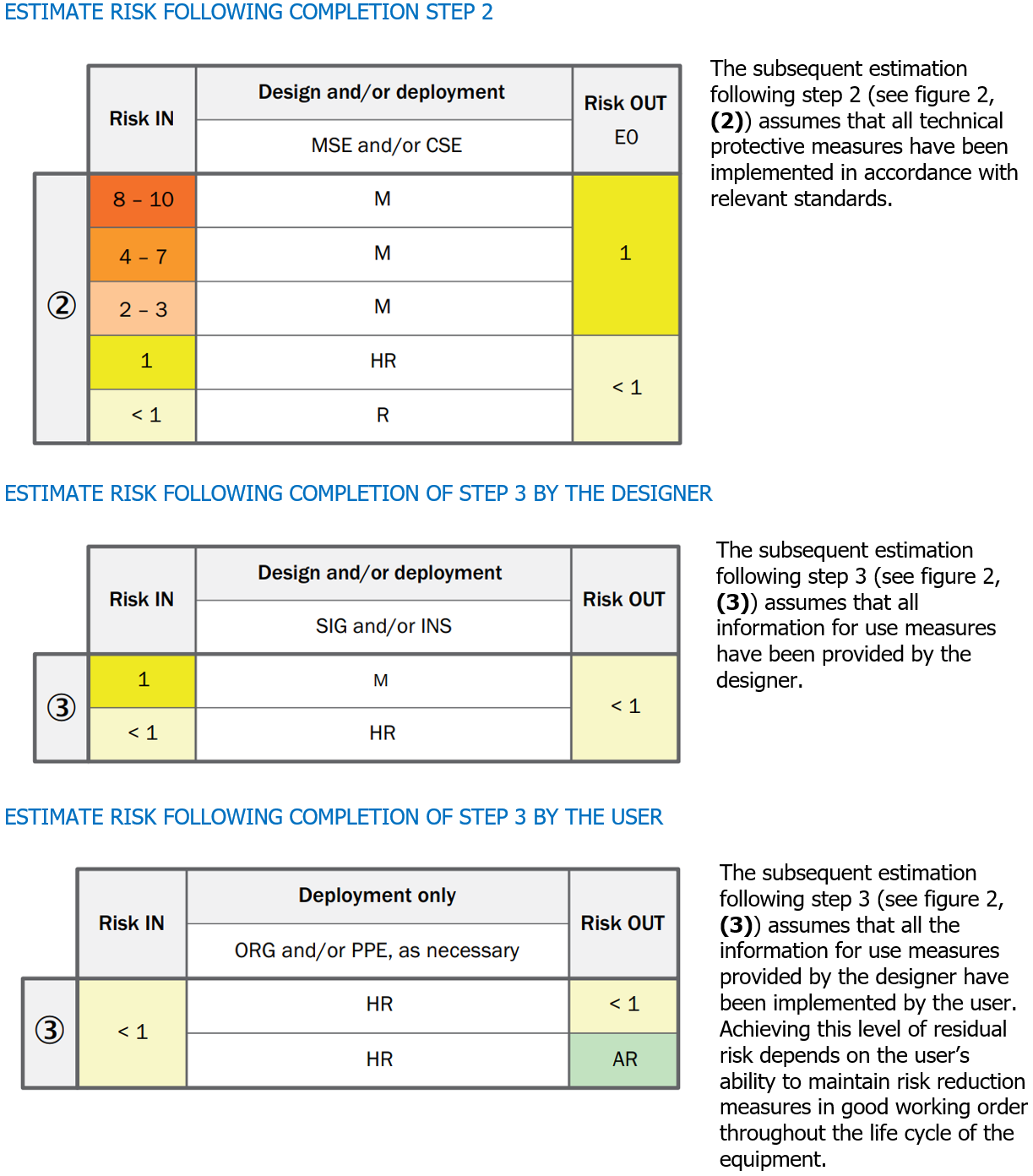
Key
|
MSE Mechanical safeguarding equipment
CSE Control-related safeguarding equipment
SIG Information at machine (e.g., signal or signs)
INS Information in instruction handbook
ORG Safe working procedures
PPE Personal protective equipment
|
M One or a combination of these measures is mandatory for this risk level
HR One or a combination of these measures is highly recommended for this risk level
R One or a combination of these measures is recommended for this risk level as a lower recommendation to an HR recommendation
|
[...]
ANNEX: Examples of the evaluation of harm severity, ANSI B11.0-2019 3rd edition
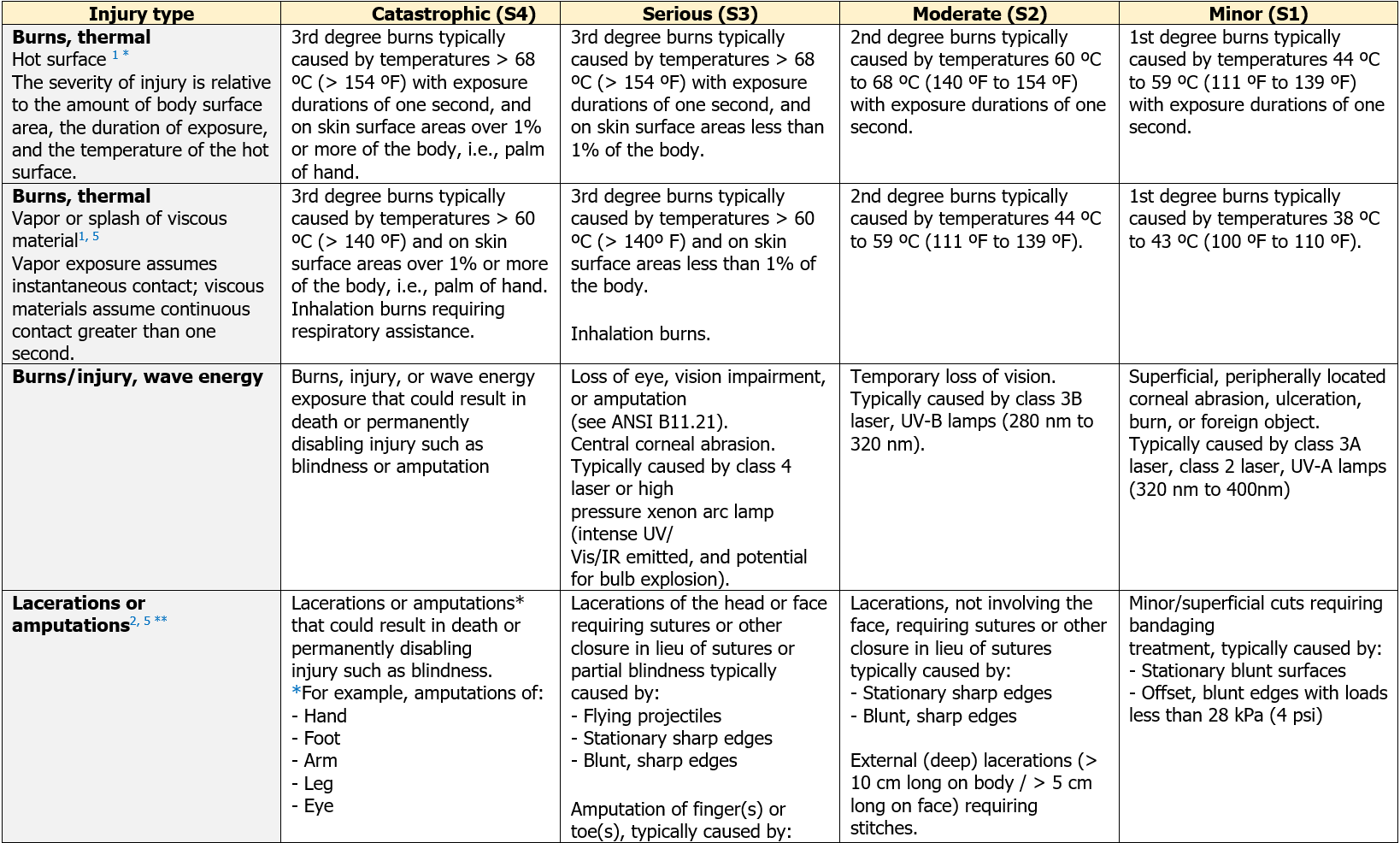
[...] Segue in allegato
Certifico Srl - EN | Rev. 1.0 2022
©Copia autorizzata Abbonati
Matrice revisioni
| Rev. |
Data |
Oggetto |
Autore |
| 1.0 |
16.09.2022 |
SICK AG - WHITE PAPER 2022-05 |
Certifico Srl |
| 0.0 |
19.01.2022 |
--- |
Certifico Srl |
Fonte: SICK
Collegati
Allegati
|
Descrizione |
Lingua |
Dimensioni |
Downloads |
 |
|
EN |
2400 kB |
95 |
 |
|
EN |
2553 kB |
256 |







































