~ 2000 / 2026 ~
// Documenti disponibili n:
47.773
// Documenti scaricati n:
38.974.514
// Newsletter n:
2972
Direttiva PED schematizzata
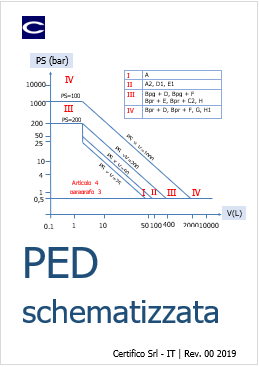
Direttiva PED schematizzata Ed. 2018
ID 7116 del 28.10.2018
Il Documento allegato illustra la Direttiva 2014/68/UE "PED", in particolare con l’aiuto di diagrammi e tabelle, si individuano le parti princ...


































