Explanatory note on MDR codes

Explanatory note on MDR codes
MDCG 2019-14 Explanatory note on MDR codes
Commission Implementing Regulation 2017/2185 establishes the codes for the designation of notified bodies in medical devices under Regulation (EU) 2017/745 and in vitro diagnostic medical devices under Regulation (EU) 2017/746.
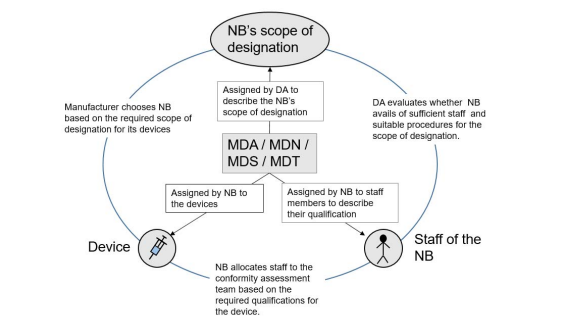
These codes are primarily used by designating authorities to define the notified body scope of designation but they are also used by the notified body to:
1) describe the individual qualification of the NBs staff members
2) describe the qualification required for assessing a device
These codes may be very broad and, furthermore, unequivocal authorisation of personnel to codes and the assignment of codes to a device is not always straightforward. However, the notified body’s system needs to ensure, in all cases, that the authorisation of personnel and team allocation for the conformity assessment of a device ensures adequate knowledge and expertise.
The lists of codes and corresponding types of devices established by the above mentioned Regulation takes into account various device types which can be characterised by design and intended purpose, manufacturing processes and technologies used, such as sterilisation and the use of nanomaterials.
These lists of codes should be used in a way that provides for a multi-dimensional application to all typology of devices. This will ensure that notified bodies as well as the staff assigned to conformity assessment are fully competent for the devices they are required to assess.
This guidance is intended to explain the different level of codes and how they should be used, including the use of conditions with a view to ensure a harmonised use of the codes especially for the allocation of resources to conformity assessment activities.
Fonte: EU
Collegati:
Allegati
|
Descrizione |
Lingua |
Dimensioni |
Downloads |
 |
|
EN |
180 kB |
8 |




































