How to apply for authorisation

How to apply for authorisation
ECHA Febbraio 2021 - Come richiedere un'autorizzazione
The purpose of this guide is to give potential applicants practical advice on how to prepare a ‘fit-for-purpose’ application for authorisation under the EU REACH Regulation, including choosing an appropriate ‘use description’. This guide is supplementary to existing ECHA guidance documents and other information on the application for authorisation process.
The European Chemicals Agency (ECHA), with input from the Task Force on the Workability of Applications for Authorisation, has prepared this guide as part of their ongoing efforts to streamline the application for authorisation process. The development of the guide was informed by feedback gathered from past applicants and other interested stakeholders and reflects the experience gained so far from the implementation of the application for authorisation process.
The guide describes the essential information that should be included in an application for authorisation and presents examples from previous applications. It identifies the important documents that applicants should familiarise themselves with before preparing an application. It also outlines some key issues that should be considered when developing an application strategy, gathering information (including supply chain communication) and application planning.
The examples from previous applications are provided to illustrate relevant issues but should not be interpreted as the best or only way of preparing an application for authorisation. Previous applications should always be read in conjunction with the corresponding opinions of the European Chemical Agency’s Scientific Committees and decisions by the European Commission as these also provide useful insight into the evaluation of applications and the decision making process.
This guide will be updated from time to time as further experience is gained in the evaluation of applications and from further understanding of the practical challenges faced by applicants. Equally, the guide will be updated in response to any improvements in the implementation of the application for authorisation process or support documentation.
This document will be revised in the future in response to improvement.
________
ECHA - Come richiedere un'autorizzazione
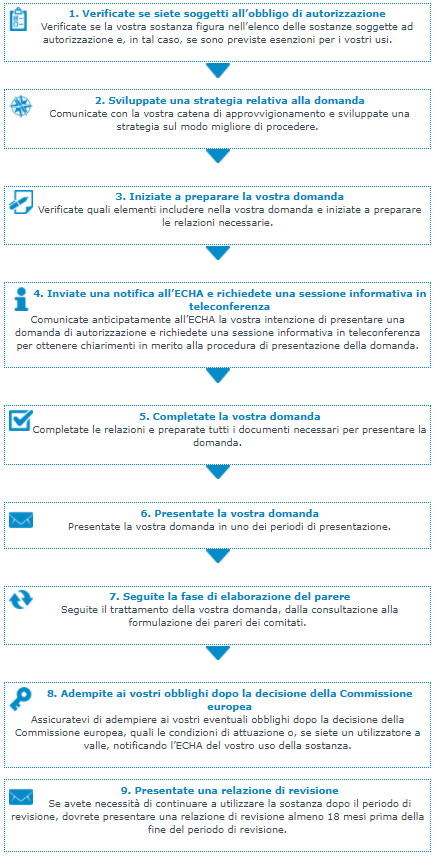
Fonte: ECHA
Collegati
Allegati
|
Descrizione |
Lingua |
Dimensioni |
Downloads |
 |
|
IT |
1452 kB |
2 |



































